Temperature & Humidity Requirements in Pharmaceutical Facilities
Temperature & Humidity Requirements in Pharmaceutical Facilities
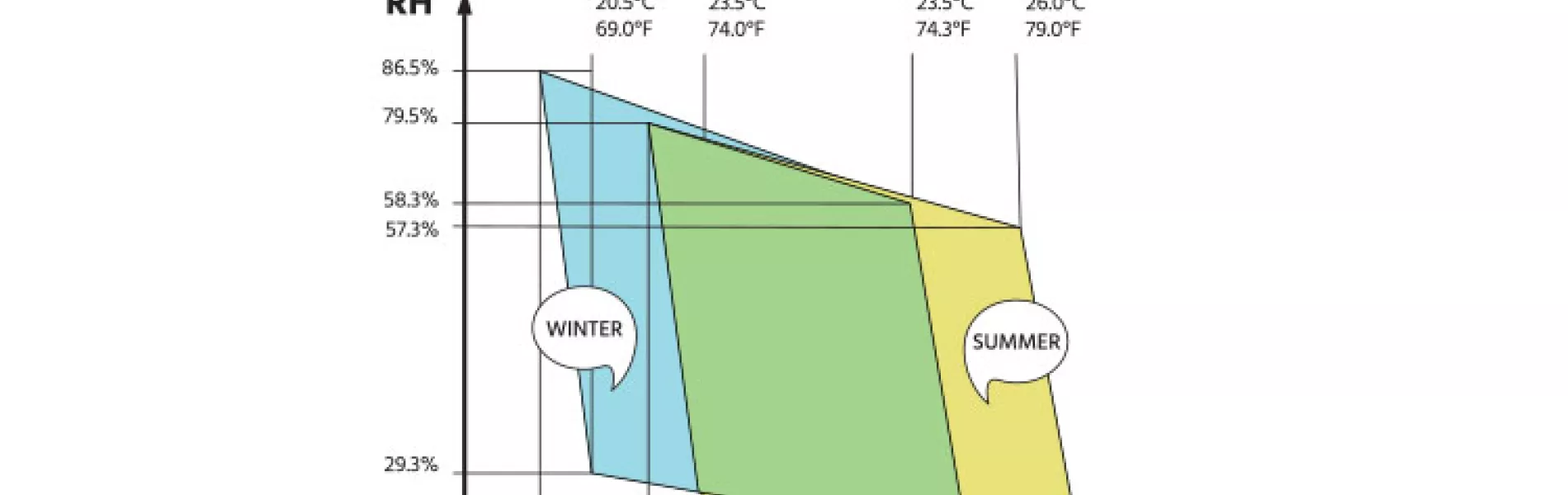
Defining room temperature and humidity limits is a frequent topic of debate when designing and operating pharmaceutical and biotechnology facilities. What are appropriate alarm limits and acceptable durations for an alarm condition? Understanding the source of temperature and humidity requirements, and strategies for setting limits, can ensure both compliance and optimum use of energy. This article provides guidance on these topics, with supporting rationales.

Pharmaceutical Graded Space Requirements

Temperature and humidity requirements in cleanroom

Supplement 6- Annex9- WHO guideline: Temperature and humidity monitoring systems

Temperature and humidity controls for textile industry

Pharmaceutical Manufacturing and Humidity Control — Humiscope

How to Investigate Temperature and Humidity Excursions of Stability Chambers American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Practical points to consider when Temperature Mapping a warehouse

Guidelines for Temperature Control of Drug Products during Storage and Transportation (GUI-0069)

Temperature Within The Pharmaceutical Industry


![Temperature and Humidity Relationship [+ Chart] - EngineerExcel Temperature and Humidity Relationship [+ Chart] - EngineerExcel](https://engineerexcel.com/wp-content/uploads/2023/03/psychrometric-chart-si-units.webp)